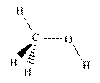Our Product : Coolant
COOLANT
Antifreeze differ from engine coolant
Engine coolant is a generic term used to describe fluids that remove heat from an engine. Antifreeze is a more specific term used to describe products used to provide protection against freezing. Many people use these terms interchangeably.
Antifreeze prevents engine’s cooling system from freezing in thewinter, what else does antifreeze do for engine’s cooling system?
Antifreeze (engine coolant) performs several functions in addition to providing freeze protection. Antifreeze contains chemicals that inhibit corrosion and scale formation in the engine and radiator. Antifreeze (engine coolant) provides protection against boiling in the summer. At one atmosphere pressure pure water boils at 212 °F (100 °C) but a 50/50 blend of water/ethylene glycol boils at 223 °F (106 °C).
Working of the cooling system
The cooling system removes excess heat from the engine block and heads, keeps the engine operating at its most efficient temperature, and gets the engine up to the correct temperature as soon as possible after starting. Ideally, the cooling system keeps the engine running at its most efficient temperature no matter what the operating conditions are.
Environmental issues require cleaner burning engines. Engine manufacturers have raised engine operating temperatures in order to reduce exhaust emissions and improve fuel economy. Today’s engines run on the borderline of overheating, with in-cylinder combustion temperatures around 2,000 °F. As fuel is burned in the engine, about one-third of the energy in the fuel is converted to kinetic energy for moving the vehicle. Another third goes out the exhaust pipe and the remaining third is removed by the cooling system. If no cooling were provided, metal parts would melt and the pistons would seize. The engine coolant must have indirect contact with the combustion chamber, the cylinder walls, and the valve seats and guides. As the engine coolant circulates through the engine, it picks up heat from the engine. The coolant releases this heat as it passes through the radiator.
Main constituents
Methanol, Ethylene glycol and Propylene glycol
Ethylene glycol
Ethylene glycol
Propylene glycol
Propylene glycol
Methanol
Methanol
Methanol, ethylene glycol and propylene glycol are chemically similar. Ethylene glycol has the chemical formula C2H6O2. Propylene has the chemical formula C3H8O2. Ethylene glycol has a slightly higher boiling point than propylene glycol. Ethylene glycol is less expensive to produce and is more widely used. Propylene glycol is less toxic.
Glycol is a petrochemical product; its price rises along with natural gas and crude oil prices. Governments are imposing more regulations concerning antifreeze management.
Propylene glycol & ethylene glycol antifreeze are environmentally friendly.
Both propylene glycol and ethylene glycol have similar biodegradability and will eventually break down into nontoxic byproducts. Neither should be dumped in the environment. Antifreeze picks up heavy metals such as lead during use in the engine. Therefore, both ethylene glycol and propylene glycol antifreezes should be returned to a recycling center to minimize harmful effects on our environment after use. Spills should be cleaned up immediately.
Ethylene glycol is more toxic than propylene glycol. Ingesting a small amount of ethylene glycol is far more dangerous for small children or animals, for example, than ingesting a small amount of propylene glycol. Take appropriate steps to prevent the ingestion of either ethylene or propylene glycol, as well as new or used engine coolants containing these glycols.
Antifreeze is poisonous
New antifreeze and used antifreeze has a sweet taste that small children and animals can find attractive. If ingested, antifreeze affects the central nervous system and can cause death.
The symptoms of antifreeze poisoning include vomiting, weakness, and loss of coordination. Contact your local poison control center for first aid information.
Most antifreeze contains ethylene glycol, which is poisonous to both humans and animals when ingested in small amounts.
Some types of antifreeze use less toxic propylene glycol, but it can still cause harm if swallowed.
Always keep antifreeze securely stored to protect yourself and others. Toxic materials such as lead and benzene might be found in automotive cooling systems. These can also be present in used antifreeze
Pure ethylene glycol is toxic to aquatic and land based life forms. It will eventually break down into water and carbon dioxide.
Used antifreeze contains more than just ethylene/propylene glycols and water. Contaminants found in used antifreeze, such as lead and benzene, can cause serious health and environmental problems. Antifreeze manufacturers also add a variety of chemicals to antifreeze to inhibit rust and corrosion. These chemicals can endanger the environment. Large quantities of glycol can suffocate aquatic life and disrupt sewage treatment processes
Propylene glycol mix with ethylene glycol
Ethylene glycol and propylene glycol are chemically very similar and can be mixed without harming the cooling system. Ethylene glycol does have better heat transfer properties than propylene glycol. Adding propylene glycol does not make the ethylene glycol less toxic.
Cooling system problems cause engine failures?
Cooling system neglect is the leading cause of premature engine failure.
Using right antifreeze
Using the right antifreeze is only part of the solution. Poor water quality and incorrect dilution are root causes for most cooling system problems
Water mix with antifreeze
Mix antifreeze with distilled water at the ratio of one part antifreeze to one part water, freeze protection down to –34°F and boil-over protection up to 265°F (if using 15 PSI pressure cap). Never use concentrated antifreeze in a cooling system without adding water. At least 40% of the mixture should be water.
Using “hard” water out of the tap can cause scaling in the engine. Tap water is purified for drinking by chlorination, which kills germs but can cause corrosion in the engine. Even if you carefully measure the amounts of tap water and antifreeze to get the right blend, using tap water is not a good idea. Tap water also contains dissolved oxygen, calcium, magnesium, and other contaminates besides chlorine and chlorides that can significantly degrade corrosion inhibitor performance
Proper water/glycol mixture.
A hydrometer uses floats to measure the specific gravity of the glycol/water mixture and indicate the amount of glycol. A refractometer measures the bending of light by the glycol/water mixture to determine the amount of glycol. Refractometers generally are more accurate than hydrometers. Measuring and mixing the water and antifreeze before adding them to the radiator is good practice. Mixing errors and the harmful effects of using tap water can be eliminated by using a pre-mixed antifreeze formulation
Most vehicle and antifreeze manufacturers recommend using pre-diluted ready-to-use antifreeze
Radiators in today’s vehicles are extremely fragile and lightweight. They contain smaller cooling tubes and more cooling fins than older models so it is easy to clog the radiators with corrosion products if you do not use the right mixture of antifreeze and water.
More than 37% of motorists jeopardize their vehicle’s cooling system by pouring either straight antifreeze or water into their radiator.

Buying premix antifreeze
Paying for high priced water
Engine manufacturers recommend using 50% water to antifreeze by volume. They also recommend that the water should be pure water, such as deionized or distilled water.
People commonly add tap water (which contains minerals, dissolved oxygen and chlorine) to concentrated antifreeze. These chemicals can quickly use up the corrosion inhibitor additives in the antifreeze. Furthermore, more often than not the final engine coolant (antifreeze/water) mix is not within the recommended range, dramatically shortening engine and cooling system life.
Not all water is created equal. Deionized or distilled water contains fewer undesirable chemicals and minerals, which results in optimum coolant life and performance. Deionized water is recommended for shop blending, but prediluted coolant is the best choice (preferred) because it ensures quality, saves time, and eliminates mixing mistakes.
If you’re interested in protecting your vehicle investment, examine some facts:
- • Antifreeze/engine coolant is the most neglected fluid in the vehicle.
- • Cooling system neglect is cited as the principal reason for premature engine and transmission failure.
- • Cooling system failure is the most common cause of mechanical breakdown on the road.
- • A national survey found that 7 out of 10 vehicles contain rust and scale and two thirds of more than 8,000 cooling system repair jobs were performed on an emergency basis.
- • Just 1/16th of an inch of mineral deposits on 1 inch of cast iron reduces heat dissipation by 40%.
A great deal of work and expense goes into the formulation of quality antifreeze/engine coolant. Reputable companies use high quality glycols, deionized or distilled water, and inhibitors that are certified to be low in contaminants (like chlorides) to blend and make premium quality finished products. Unfortunately, these efforts are wasted when consumers use unsuitable tap water to self-blend the antifreeze.
Calcium and Magnesium – The total concentration of these elements is called water hardness. Calcium and magnesium salts form scale on hot heat exchange surfaces. Scale impedes heat transfer and causes localized hot spots those results in engine overheating and component failure.
Chloride and Chlorine – All municipal water supplies contain chloride and chlorine. Chloride is very corrosive to all cooling system metals, especially aluminum. Chlorine forms additional chlorides in the cooling system.
Sulfate – All municipal water also contains sulfate that contributes to general corrosion and scale formation.
Oxygen – Most people don’t drink de-ionized water because they do not like its taste. De-ionized water doesn't taste good due to the lack of oxygen and certain minerals. Tap water, however, is full of oxygen and other minerals making it suitable for drinking and aquariums. Oxygen contributes to metal corrosion and depletion of inhibitors. Water with low oxygen is preferred for engine coolant.
Mixing mistakes - Cooling system problems can also be caused by improper dilution of the coolant/antifreeze with water. These problems include:
- • Improper ratio of concentrate to water
- • Not stirring thoroughly to mix the water and concentrate
- • Mixing the concentrate and water in a dirty container
- • Pouring the concentrate and water into the radiator without first mixing them

Header scale - Consequence of using tap water
So if you have used tap water in your antifreeze, you are overdue for cooling system maintenance because deposits likely have been building up. Maybe you already have had the radiator corroded out. Unfortunately, the engine block and heads have the same deposits. A quick acid flush (which can attack copper, aluminum, and brass) will NOT remove what took years and tens of thousands of miles to build up.